 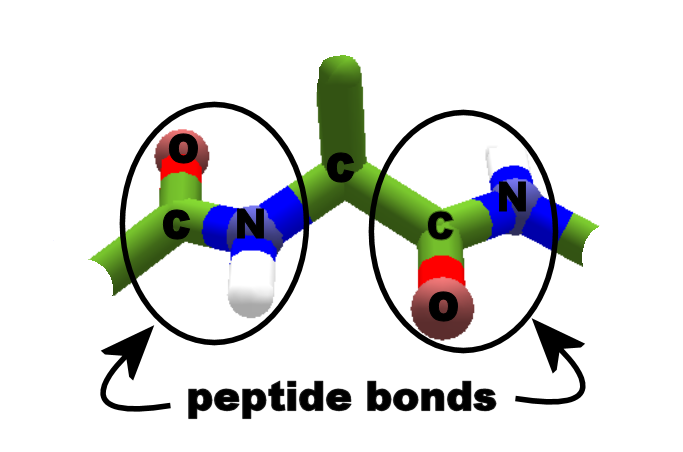 
However, the H-bonding residues are present in adjacently opposed stretches of the polypeptide backbone as opposed to a linearly contiguous region of the backbone in the α-helix. The folding and alignment of stretches of the polypeptide backbone aside one another to form β-sheets is stabilized by H-bonding between amide nitrogens and carbonyl carbons. Whereas an α-helix is composed of a single linear array of helically disposed amino acids, β-sheets are composed of 2 or more different regions of stretches of at least 5-10 amino acids. The disruption of the helix is important as it introduces additional folding of the polypeptide backbone to allow the formation of globular proteins. This is particularly true for P since it is a pyrrolidine based imino acid (HN=) whose structure significantly restricts movement about the peptide bond in which it is present, thereby, interfering with extension of the helix. Amino acids such as A, D, E, I, L and M favor the formation of α-helices, whereas, G and P favor disruption of the helix. Not all amino acids favor the formation of the (α-helix due to steric constraints of the R-groups. This orientation of H-bonding produces a helical coiling of the peptide backbone such that the R-groups lie on the exterior of the helix and perpendicular to its axis.īall and stick model of an alpha helix in protein The formation of the α-helix is spontaneous and is stabilized by H-bonding between amide nitrogens and carbonyl carbons of peptide bonds spaced four residues apart. The α-helix is a common secondary structure encountered in proteins of the globular class. Within a single protein different regions of the polypeptide chain may assume different conformations determined by the primary sequence of the amino acids. It is the partial double-bond character of the peptide bond that defines the conformations a polypeptide chain may assume. Globular proteins are compactly folded and coiled, whereas, fibrous proteins are more filamentous or elongated. In general proteins fold into two broad classes of structure termed, globular proteins or fibrous proteins. These conformations constitute the secondary structures of a protein. The ordered array of amino acids in a protein confer regular conformational forms upon that protein. the end with the residue containing a free α-carboxyl group) is to the right. the end bearing the residue with the free α-amino group) is to the left (and the number 1 amino acid) and the C-terminal end (i.e. The convention for the designation of the order of amino acids is that the N-terminal end (i.e. The primary structure of peptides and proteins refers to the linear number and order of the amino acids present. High Performance Liquid Chromatography (HPLC).Table of the Specificities of Several Exopeptidases.Carboxy-Terminal Sequence Determination.Table of the Specificities of Several Endoproteases.Analysis of Protein Structure and Composition. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
